The 4M Laboratory investigates the biomechanical origins and progression of knee osteoarthritis (OA) through complementary computational and experimental approaches. Our work bridges the gap between molecular-scale tissue degradation and whole-joint mechanics, with the goal of informing patient-specific treatment and surgical planning strategies. Current efforts focus on ACL reconstruction outcomes, cartilage degradation mechanisms mediated by proteases such as MMPs and ADAMTS, and multiscale finite element models that couple musculoskeletal dynamics with soft tissue mechanics.
Anterior cruciate ligament (ACL) re-constructive surgeries are often successful at improving joint stability, but patients have a highly elevated risk for developing early onset osteoarthritis (OA). It is not well understood why OA develops in these patients, or how one can best plan a treatment strategy to mitigate the risk for OA.
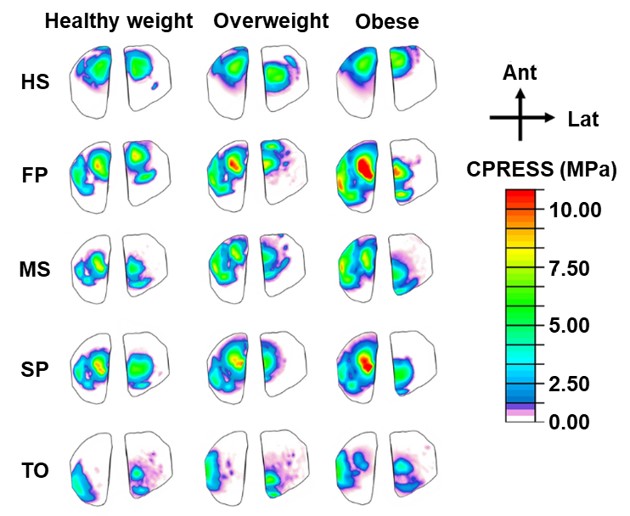
A new multiscale computational construct is being formulated to examine the inherent coupling that exists between musculoskeletal dynamics and soft tissue mechanics, which are decoupled in existing computational models, during gait. This construct is used to gain insights into the key surgical factors that can affect cartilage contact stresses after ACL reconstruction, which are believed to predicate osteoarthritis changes to the joint.
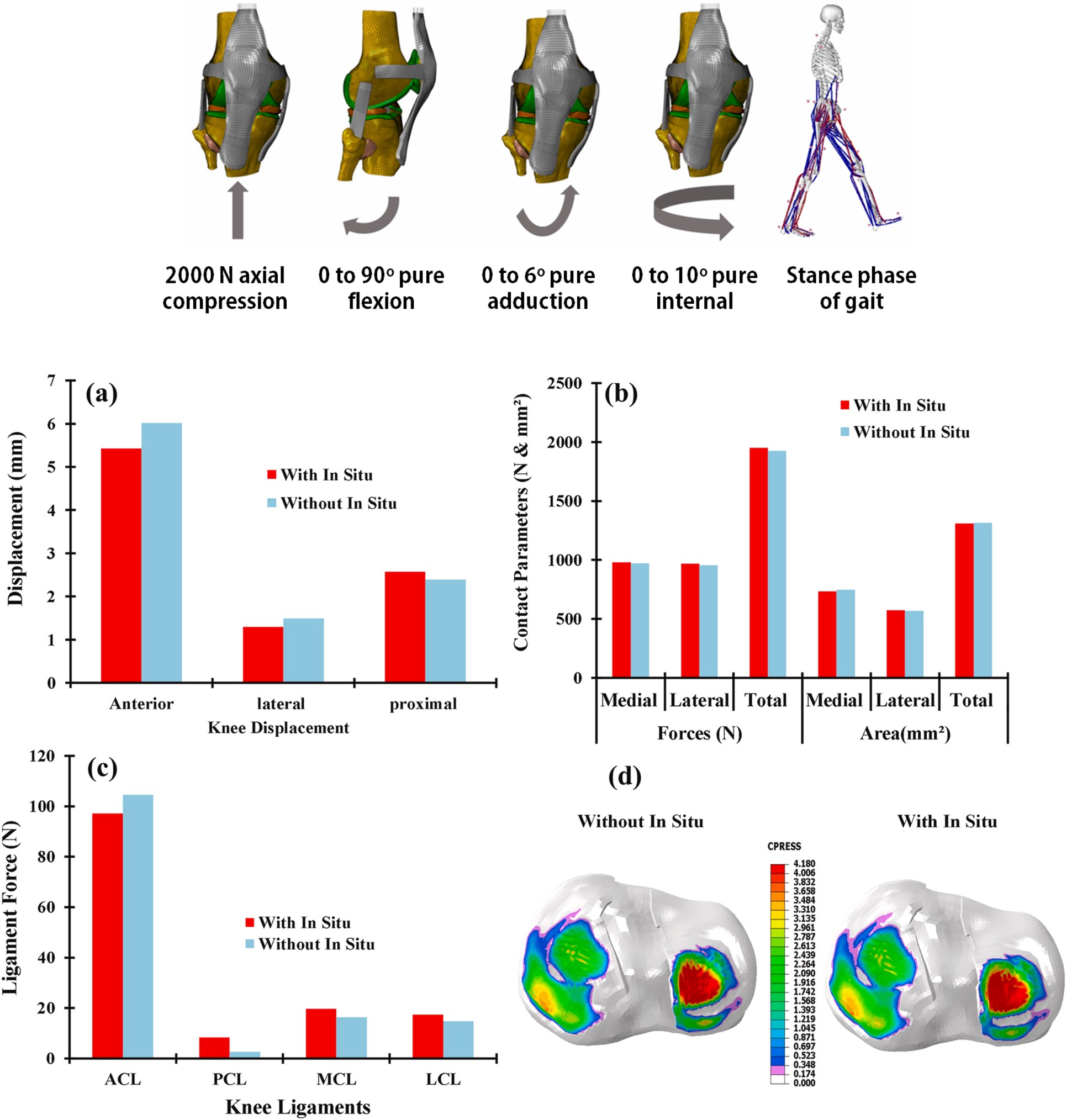
The aims of this research are to investigate the coupled influence of surgical factors and muscle forces on in vivo knee mechanics as measured using dynamic MRI and to investigate the multiscale biomechanical behavior of reconstructed knees during human locomotion as well. The results of these studies are important for informing clinical approaches that can best mitigate the risk of OA following ligament injury and surgical repair.
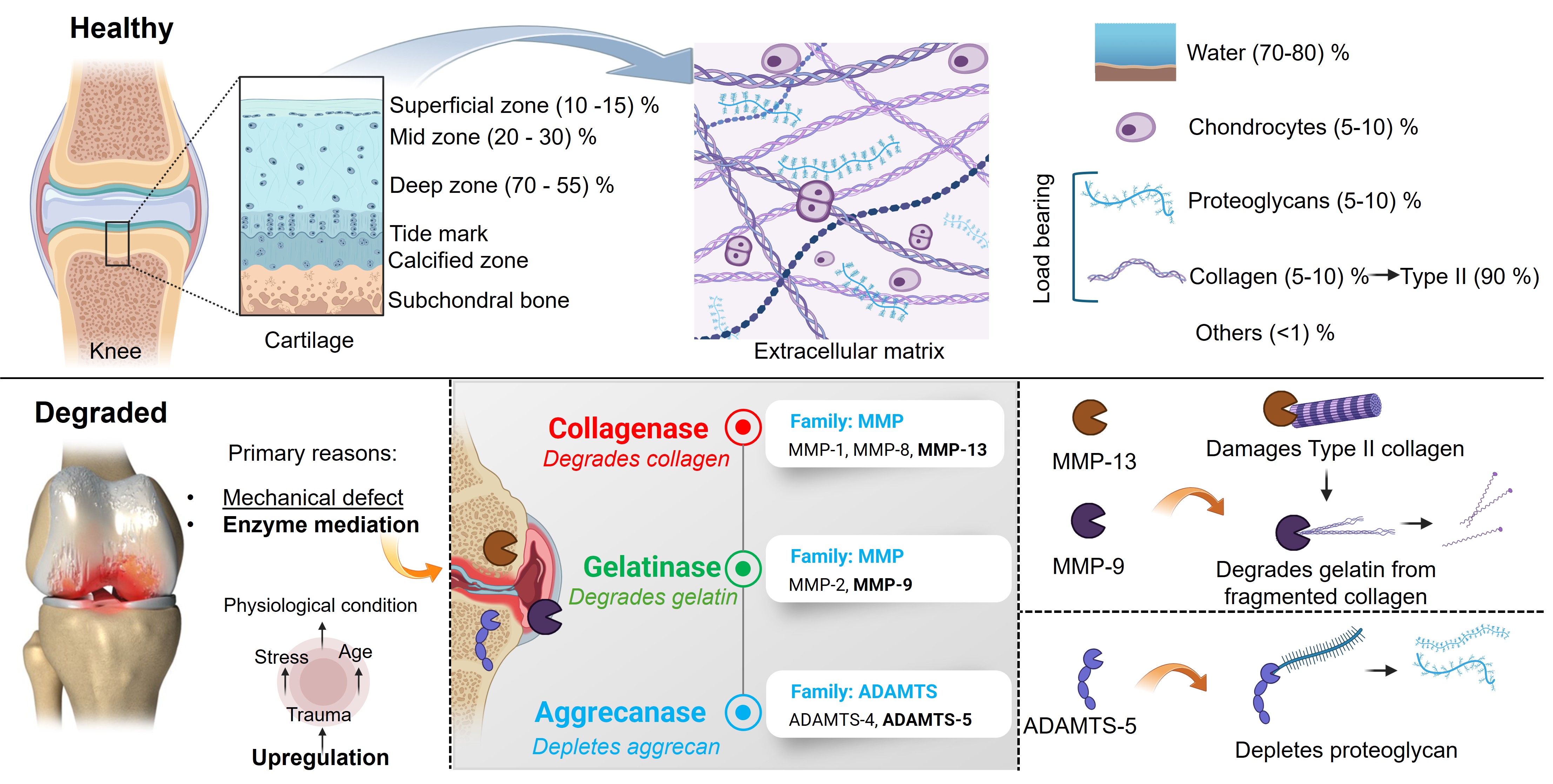
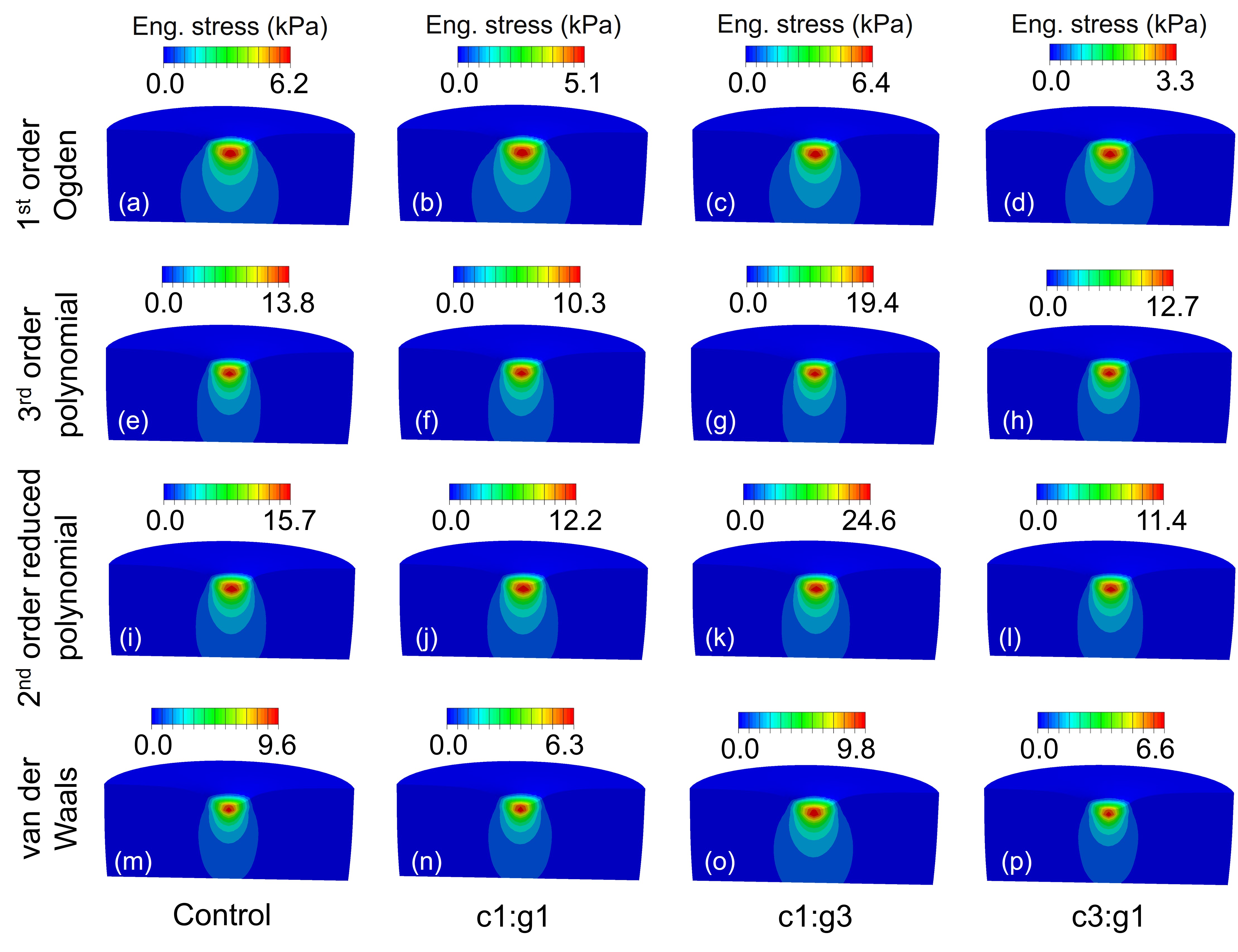
Parallel to our computational work, the 4M Laboratory conducts experimental investigations to characterize articular cartilage tissue mechanics across healthy and degraded states. Using bovine and human cartilage specimens, we apply controlled enzymatic degradation protocols — specifically co-incubation with MMP-1 and MMP-9, the collagenase and gelatinase isoforms implicated in early OA — to produce graded tissue damage in vitro. Mechanical testing via indentation and unconfined compression quantifies changes in elastic modulus, permeability, and viscoelastic response as a function of degradation severity. Our in vitro studies established that MMP-1 and MMP-9 act synergistically, producing significantly greater reductions in tissue stiffness than either enzyme alone. These findings motivated subsequent work on therapeutic inhibition, in which we demonstrated that exogenous COMP and TIMP-3 can partially restore cartilage mechanical integrity compromised by protease activity — validated through combined histomorphometric and mechanical analysis . These experimental findings directly calibrate and validate the constitutive models used in our computational simulations.